
Chemistry, 30.03.2020 21:06 annagwhiteou0hrh
Calculate the standard entropy change for the reaction: 2 hydrogen sulfide(g) plus sulfur dioxide (g) goes to form 3 sulfur(s) plus 2 water(g). Given S(s) = 31.88 J/mol. Look up the other values in the text appendix. No units required. Just the numerical answer

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
What is the value of keq for the reaction expressed in scientific notation
Answers: 1

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3


Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1
You know the right answer?
Calculate the standard entropy change for the reaction: 2 hydrogen sulfide(g) plus sulfur dioxide (g...
Questions

Mathematics, 12.11.2020 02:10

Biology, 12.11.2020 02:10




Business, 12.11.2020 02:10



English, 12.11.2020 02:10

History, 12.11.2020 02:10



Mathematics, 12.11.2020 02:10


Mathematics, 12.11.2020 02:10

English, 12.11.2020 02:10


Chemistry, 12.11.2020 02:10


Chemistry, 12.11.2020 02:10

![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0571/2033/52737.png)
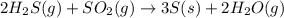
![\Delta S^o_{rxn}=[(3\times \Delta S^o_{(S(s))})+(2\times \Delta S^o_{(H_2O(g))})]-[(2\times \Delta S^o_{(H_2S(g))})+(1\times \Delta S^o_{(SO_2(g))})]](/tpl/images/0571/2033/492b6.png)
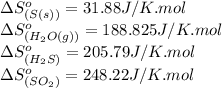
![\Delta S^o_{rxn}=[(3\times (31.88))+(2\times (188.825))]-[(2\times (205.79))+(1\times (248.22))]\\\\\Delta S^o_{rxn}=-186.51J/K](/tpl/images/0571/2033/7cc5d.png)


