
Chemistry, 30.03.2020 21:22 dpazmembreno
A 35.0 L sample of gas collected in the upper atmosphere at a pressure of 48.6 torr is compressed into a 150. mL container at constant temperature. What is the new pressure (atm)? (4pts) 1 atm = 760 torr

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3
You know the right answer?
A 35.0 L sample of gas collected in the upper atmosphere at a pressure of 48.6 torr is compressed in...
Questions


Mathematics, 09.10.2019 01:50

Mathematics, 09.10.2019 01:50


Chemistry, 09.10.2019 01:50

History, 09.10.2019 01:50


History, 09.10.2019 01:50

History, 09.10.2019 01:50

Health, 09.10.2019 01:50





Mathematics, 09.10.2019 01:50


Mathematics, 09.10.2019 02:00

Mathematics, 09.10.2019 02:00


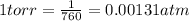
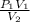
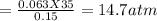 or 11172 torr
or 11172 torr 


