
Chemistry, 30.03.2020 22:42 OGrant18075
Following the instructions in your lab manual, you have titrated a 25.00 mL sample of 0.0100 M KIO3 with a solution of Na2S2O3 of unknown concentration. The endpoint was observed to occur at 16.50 mL. 1. How many moles of KIO3 were titrated? Show work! 2. How many moles of Na2S2O3 did this require?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 08:20
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2
You know the right answer?
Following the instructions in your lab manual, you have titrated a 25.00 mL sample of 0.0100 M KIO3...
Questions

Mathematics, 10.02.2022 16:40



Mathematics, 10.02.2022 16:40



Mathematics, 10.02.2022 16:40


Mathematics, 10.02.2022 16:50



Advanced Placement (AP), 10.02.2022 16:50

Mathematics, 10.02.2022 16:50

Computers and Technology, 10.02.2022 16:50

Mathematics, 10.02.2022 16:50


English, 10.02.2022 16:50



Mathematics, 10.02.2022 16:50

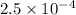 moles
moles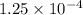 moles
moles
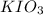 solution = 0.0100 M
solution = 0.0100 M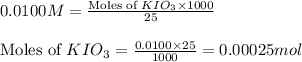
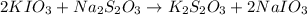
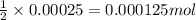 of sodium thiosulfate
of sodium thiosulfate

