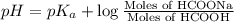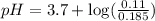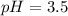Chemistry, 30.03.2020 23:33 erikamaldonado661
Calculate the pH of a solution that contains 0.250 M formic acid, HCOOH (Ka =1.8 x 10-4 ), and 0.100M sodium formate, HCOONa after the addition of 10.0 mL of 6.00M NaOH to the original buffered solution volume of 500.0 mL.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2
You know the right answer?
Calculate the pH of a solution that contains 0.250 M formic acid, HCOOH (Ka =1.8 x 10-4 ), and 0.100...
Questions


Mathematics, 11.01.2021 16:20

Biology, 11.01.2021 16:20





Computers and Technology, 11.01.2021 16:20

English, 11.01.2021 16:20





English, 11.01.2021 16:20



Chemistry, 11.01.2021 16:20



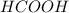 ,
, 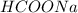 and
and 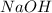 .
.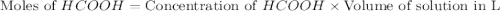
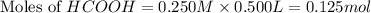
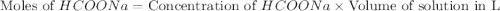
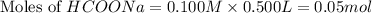
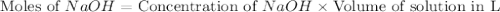
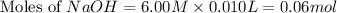
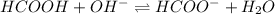
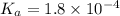
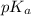 .
.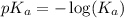
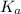 in this expression, we get:
in this expression, we get: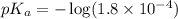
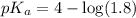
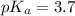
![pH=pK_a+\log \frac{[Salt]}{[Acid]}](/tpl/images/0571/8655/e961a.png)
![pH=pK_a+\log \frac{[HCOONa]}{[HCOOH]}](/tpl/images/0571/8655/c5edc.png)
![pH=pK_a+\log \frac{[\frac{\text{Moles of HCOONa}}{\text{Volume of solution}}]}{[\frac{\text{Moles of HCOOH}}{\text{Volume of solution}}]}](/tpl/images/0571/8655/fba20.png)