
Chemistry, 31.03.2020 01:07 donttrip10
Consider the reaction: 2 Al + 3Br2 → 2 AlBr3 Suppose a reaction vessel initially contains 5.0 mole Al and 6.0 mole Br2. What is in the reaction vessel once the reaction has occurred to the fullest extent possible? chapter 9

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
Consider the reaction: 2 Al + 3Br2 → 2 AlBr3 Suppose a reaction vessel initially contains 5.0 mole A...
Questions


Biology, 24.08.2019 03:00

French, 24.08.2019 03:00



History, 24.08.2019 03:00

Physics, 24.08.2019 03:00

Mathematics, 24.08.2019 03:00

English, 24.08.2019 03:00

History, 24.08.2019 03:00


English, 24.08.2019 03:00



Biology, 24.08.2019 03:00


Arts, 24.08.2019 03:00


Mathematics, 24.08.2019 03:00

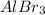 and 1 mole of
and 1 mole of 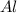 will be present in the reaction vessel
will be present in the reaction vessel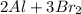 ⇒
⇒ 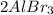
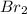 so in this reaction,
so in this reaction, 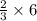 = 4 moles.
= 4 moles.

