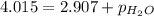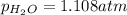Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
You know the right answer?
A cylinder contains nitrogen and oxygen under 4.015 atm pressure. The partial pressure of N2 is 2.90...
Questions


History, 01.12.2020 02:10


Engineering, 01.12.2020 02:10

Health, 01.12.2020 02:10


Mathematics, 01.12.2020 02:10


Advanced Placement (AP), 01.12.2020 02:10

Mathematics, 01.12.2020 02:10

Mathematics, 01.12.2020 02:10


Health, 01.12.2020 02:10


Mathematics, 01.12.2020 02:10


Mathematics, 01.12.2020 02:10



Business, 01.12.2020 02:10

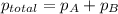
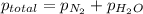
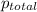 =4.015 atm
=4.015 atm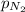 = 2.907 atm
= 2.907 atm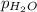 = ?
= ?