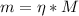Chemistry, 31.03.2020 04:40 Gogontle8347
A chemist must prepare 800.0mL of sodium hydroxide solution with a pH of 12.10 at 25°C. She will do this in three steps: Fill a 800.0mL volumetric flask about halfway with distilled water. Weigh out a small amount of solid sodium hydroxide and add it to the flask. Fill the flask to the mark with distilled water. Calculate the mass of sodium hydroxide that the chemist must weigh out in the second step. Round your answer to 2 significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
A chemist must prepare 800.0mL of sodium hydroxide solution with a pH of 12.10 at 25°C. She will do...
Questions

Chemistry, 30.08.2019 02:30


Biology, 30.08.2019 02:30

Health, 30.08.2019 02:30

Computers and Technology, 30.08.2019 02:30



History, 30.08.2019 02:30









Mathematics, 30.08.2019 02:30

Chemistry, 30.08.2019 02:30



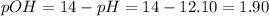
![pOH = -log ([OH^{-}])](/tpl/images/0572/8536/5638e.png)
![[OH]^{-} = 10^{-pOH} = 10^{-1.90} = 0.013 M](/tpl/images/0572/8536/7c501.png)
![\eta = ([OH]^{-})*V = 0.013 mol/L * 0.800 L = 1.04 \cdot 10^{-2} moles](/tpl/images/0572/8536/c1790.png)