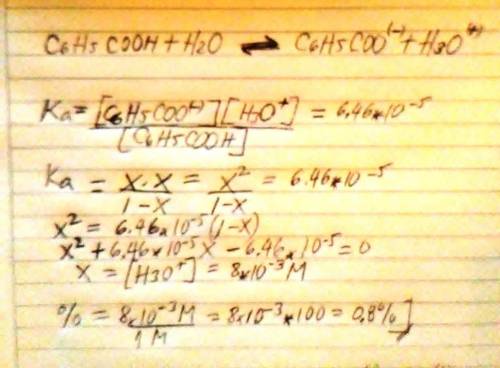The acid ionization constant for benzoic acid (C6H5COOH) is 6.46 × 10−5 . Compare the percent ionization of 1.00 M benzoic acid in water with its percent ionization in 0.500 M sodium benzoate solution. Support your comparison with calculations similar to those in Model 1.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1
You know the right answer?
The acid ionization constant for benzoic acid (C6H5COOH) is 6.46 × 10−5 . Compare the percent ioniza...
Questions







Mathematics, 22.01.2021 20:00

English, 22.01.2021 20:00

Chemistry, 22.01.2021 20:00

Chemistry, 22.01.2021 20:00

Advanced Placement (AP), 22.01.2021 20:00

Mathematics, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00




Social Studies, 22.01.2021 20:00