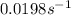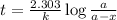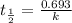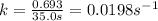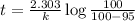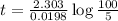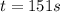Chemistry, 02.04.2020 03:27 cuddleslugsovifat
Be sure to answer all parts. The thermal decomposition of phosphine (PH3) into phosphorus and molecular hydrogen is a first-order reaction: 4PH3(g) → P4(g) + 6H2(g) The half-life of the reaction is 35.0 s at 680°C.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1
You know the right answer?
Be sure to answer all parts. The thermal decomposition of phosphine (PH3) into phosphorus and molecu...
Questions


Mathematics, 17.07.2019 04:30




Mathematics, 17.07.2019 04:30


History, 17.07.2019 04:30


Biology, 17.07.2019 04:30





Mathematics, 17.07.2019 04:30


History, 17.07.2019 04:30

Mathematics, 17.07.2019 04:30


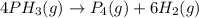 The half-life of the reaction is 35.0 s at 680°C. Calculate the first order rate constant.
The half-life of the reaction is 35.0 s at 680°C. Calculate the first order rate constant.