
Chemistry, 04.04.2020 02:33 nate102201
Potassium hydroxide is very soluble in water, resulting in extremely basic solutions. A 121g sample KOH is dissolved in water at 25∘C to make up 100.0mL of solution. The molar mass of KOH is 56.11gmol. What is the pH of the solution at 25.0∘C?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
Potassium hydroxide is very soluble in water, resulting in extremely basic solutions. A 121g sample...
Questions

Biology, 19.07.2019 01:30



Mathematics, 19.07.2019 01:30

Geography, 19.07.2019 01:30


English, 19.07.2019 01:30


Mathematics, 19.07.2019 01:30

Mathematics, 19.07.2019 01:30





Mathematics, 19.07.2019 01:30


Mathematics, 19.07.2019 01:30

Health, 19.07.2019 01:30

Spanish, 19.07.2019 01:30

Chemistry, 19.07.2019 01:30

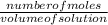
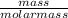 =
= 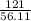 = 2.16mole
= 2.16mole 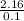 = 21.6moldm⁻³
= 21.6moldm⁻³

