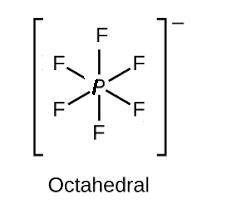
According to VSEPR theory, the molecule PF6- has how many regions of electron density around the central atom (how many electron groups?) (enter a number from 1 to 6) PF6- will have (linear, trigonal planar, tetrahedral, trigonal pyramidal, bent, trigonal bipyramidal, seesaw, T-shaped, octahedral, square pyramidal, square planar) electronic geometry, and (linear, trigonal planar, tetrahedral, trigonal pyramidal, bent, trigonal bipyramidal, seesaw, T-shaped, octahedral, square pyramidal, square planar) molecular geometry.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 23.06.2019 10:00
Which element forms a compound with chlorine with the general formula mci?
Answers: 1
You know the right answer?
According to VSEPR theory, the molecule PF6- has how many regions of electron density around the cen...
Questions

English, 01.08.2021 17:00

Social Studies, 01.08.2021 17:00

Social Studies, 01.08.2021 17:00




Mathematics, 01.08.2021 17:00


Mathematics, 01.08.2021 17:00


Social Studies, 01.08.2021 17:00


Arts, 01.08.2021 17:00

Physics, 01.08.2021 17:00

Mathematics, 01.08.2021 17:10

Geography, 01.08.2021 17:10




![\text{Number of electron pair}=\frac{1}{2}[V+N-C+A]](/tpl/images/0582/2287/9e987.png)
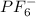
![\text{Number of electrons}=\frac{1}{2}\times [5+6+1]=6](/tpl/images/0582/2287/ae23d.png)
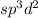 and the electronic geometry and the molecular geometry of the molecule will be octahedral.
and the electronic geometry and the molecular geometry of the molecule will be octahedral.