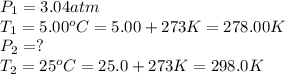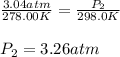Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1

You know the right answer?
Air was put into an automobile tire when the tire was cold, at 5.00 C. The tire’s air pressure was 3...
Questions




Health, 05.09.2019 20:30

History, 05.09.2019 20:30





History, 05.09.2019 20:30

Biology, 05.09.2019 20:30

Biology, 05.09.2019 20:30


Mathematics, 05.09.2019 20:30



Mathematics, 05.09.2019 20:30

Advanced Placement (AP), 05.09.2019 20:30


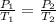
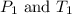 are the initial pressure and temperature of the gas.
are the initial pressure and temperature of the gas.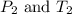 are the final pressure and temperature of the gas.
are the final pressure and temperature of the gas.