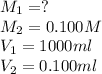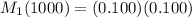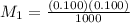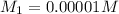Chemistry, 05.04.2020 03:01 maddie7155
A 1000.0 ml sample of lake water in titrated using 0.100 ml of a 0.100 M base solution. What is the molarity (M) of the acid in the lake water ?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
A 1000.0 ml sample of lake water in titrated using 0.100 ml of a 0.100 M base solution. What is the...
Questions







Mathematics, 08.01.2020 07:31

Mathematics, 08.01.2020 07:31

Mathematics, 08.01.2020 07:31

History, 08.01.2020 07:31

Chemistry, 08.01.2020 07:31

Biology, 08.01.2020 07:31


Arts, 08.01.2020 07:31

Computers and Technology, 08.01.2020 07:31


History, 08.01.2020 07:31


History, 08.01.2020 07:31

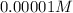 .
.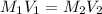
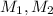 are molarity of acid in the lake water and base solution respectively.
are molarity of acid in the lake water and base solution respectively.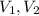 are volume of sample in the lake water and base solution respectively.
are volume of sample in the lake water and base solution respectively.