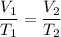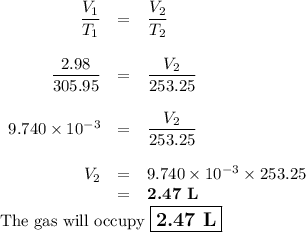Chemistry, 05.04.2020 20:47 viviansotelo12
8.98L of gas is collected at 32.8°C. Find the volume the gas will occupy at -19.9°C if the pressure remains constant.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 01:40
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
8.98L of gas is collected at 32.8°C. Find the volume the gas will occupy at -19.9°C if the pressure...
Questions




Chemistry, 12.06.2020 18:57

Mathematics, 12.06.2020 18:57

Mathematics, 12.06.2020 18:57



Spanish, 12.06.2020 18:57


Mathematics, 12.06.2020 18:57

Advanced Placement (AP), 12.06.2020 18:57

English, 12.06.2020 18:57

English, 12.06.2020 18:57


Mathematics, 12.06.2020 18:57



Mathematics, 12.06.2020 18:57

Computers and Technology, 12.06.2020 18:57