Chemistry, 06.04.2020 23:45 markarianlaura1
2- A sheet of steel 3.0-mm thick has nitrogen atmospheres on both sides at 900°C and is permitted to achieve a steady-state diffusion condition. The diffusion coefficient for nitrogen in steel at this temperature is 1.85 à 10â10 m2/s, and the diffusion flux is found to be 1.0 à 10â7 kg/m2 . s. Also, it is known that the concentration of nitrogen in the steel at the high-pressure surface is 2 kg/m3. How far into the sheet from this high-pressure side will the concentration be 0.5 kg/m3? Assume a linear concentration profile. (40 pts.)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is in the lanthanide family? a) uranium b) promethium c) silver d) gold
Answers: 2
You know the right answer?
2- A sheet of steel 3.0-mm thick has nitrogen atmospheres on both sides at 900°C and is permitted t...
Questions

Business, 06.10.2021 01:00

History, 06.10.2021 01:00











Biology, 06.10.2021 01:00


Mathematics, 06.10.2021 01:00





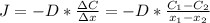
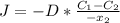 (1)
(1)