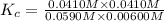Chemistry, 07.04.2020 03:15 sethlynn2003
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the concentrations of the substances at equilibrium.
Use the
appropriate significant figures in reporting the answers.
CO(g) + H2O(g) ⇌ CO2(g) + H2(g) [CO] = 0.0590 M; [H2O] = 0.00600 M;
[CO2] = 0.0410 M; [H2] = 0.0410 M
K =-

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 13:00
Imagine that you push on a large rock. at what point does your effort change the rock’s mechanical energy?
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the con...
Questions


Mathematics, 07.01.2020 01:31

Mathematics, 07.01.2020 01:31

Social Studies, 07.01.2020 01:31

Mathematics, 07.01.2020 01:31

Mathematics, 07.01.2020 01:31

Mathematics, 07.01.2020 01:31

Mathematics, 07.01.2020 01:31


History, 07.01.2020 01:31


Mathematics, 07.01.2020 01:31

History, 07.01.2020 01:31





Mathematics, 07.01.2020 01:31


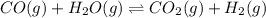
![[CO]=0.0590 M,[H_2O]=0.00600 M](/tpl/images/0585/8718/63397.png)
![[CO_2]=0.0410 M,[H_2]=0.0410 M](/tpl/images/0585/8718/efcda.png)
![K_c=\frac{[CO_2][H_2]}{[CO][H_2O]}](/tpl/images/0585/8718/fbbde.png)