
Chemistry, 07.04.2020 04:59 tnbankspines
Using your value of Ksp, and starting with an equilibrium system consisting of a saturated solution of calcium hydroxide, predict whether the solubility will increase or decrease it you add a. A. 5.00 mL of 0.100 M HNO3 Ksp = 2.966610-5 b. 5.00 mL of 0.100 M NaOH C. 5.00 mL of 0.100 M Ca(NO3)2

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Afamily is one another name for a group on the table of elements.
Answers: 1


Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2

Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer
Answers: 1
You know the right answer?
Using your value of Ksp, and starting with an equilibrium system consisting of a saturated solution...
Questions










English, 03.09.2020 01:01



Social Studies, 03.09.2020 01:01

Mathematics, 03.09.2020 01:01




Mathematics, 03.09.2020 01:01


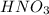 Solubility will increase.
Solubility will increase.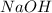 Solubility will decrease.
Solubility will decrease.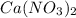 Solubility will decrease.
Solubility will decrease.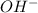 are consumed due to it and the system would respond by reaction shifting towards creation of more
are consumed due to it and the system would respond by reaction shifting towards creation of more 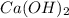 precipitating back. Hence solubility decreases.
precipitating back. Hence solubility decreases.
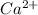 which will be countered by reaction shifting to side that decreases increased
which will be countered by reaction shifting to side that decreases increased 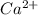 , which happens by some
, which happens by some 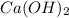 precipitating back. Hence solubility decreases.
precipitating back. Hence solubility decreases.

