

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Elements that do not have full outer electron shells will donate, share, or take electrons from other atoms. choose the items that have the correct binary ionic formula.
Answers: 2


Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3

Chemistry, 23.06.2019 10:00
Compare and contrast an assemblage and a pollen fingerprint by defying both and giving examples of each from the chapter.
Answers: 3
You know the right answer?
2) Give the number of lone pairs around the central atom and the molecular geometry of IF5. A) 0 lon...
Questions

Mathematics, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Geography, 12.12.2020 17:00

Social Studies, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Biology, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00


Social Studies, 12.12.2020 17:00



Mathematics, 12.12.2020 17:00

Chemistry, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00

![\text{Number of electrons}=\frac{1}{2}[V+N-C+A]](/tpl/images/0586/4224/86ecf.png)
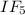 molecule.
molecule.![\text{Number of electrons}=\frac{1}{2}\times [7+5+0-0]=6](/tpl/images/0586/4224/6e043.png)
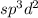 and the electronic geometry of the molecule will be octahedral .
and the electronic geometry of the molecule will be octahedral . 

