
A sample of nitrogen gas had a volume of 500. mL, a pressure in its closed container of 740 torr, and a temperature of 25 °C. What was the new volume of the gas when the temperature was changed to 50 °C and the new pressure was 760 torr, if the amount of gas does not change?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 09:10
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
A sample of nitrogen gas had a volume of 500. mL, a pressure in its closed container of 740 torr, an...
Questions

Mathematics, 02.12.2020 18:30

Social Studies, 02.12.2020 18:30



Mathematics, 02.12.2020 18:30



Mathematics, 02.12.2020 18:30

Social Studies, 02.12.2020 18:30

Mathematics, 02.12.2020 18:30

Geography, 02.12.2020 18:30

Mathematics, 02.12.2020 18:30

History, 02.12.2020 18:30

Mathematics, 02.12.2020 18:30


Law, 02.12.2020 18:30

Mathematics, 02.12.2020 18:30


Mathematics, 02.12.2020 18:30

English, 02.12.2020 18:30

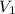 = 500 mL = 0.5 L
= 500 mL = 0.5 L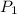 = 740 torr
= 740 torr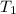 = 25 ° c = 298 K
= 25 ° c = 298 K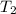 = 50 ° c = 323 K
= 50 ° c = 323 K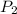 = 760 torr
= 760 torr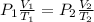
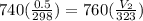
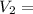 0.526 L = 526 ml
0.526 L = 526 ml

