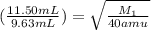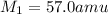If 11.50 mL of an unknown gas effuses through a small hole in a heated vessel in the same time it takes 9.63 mL of argon to effuse through the hole under the same conditions, what is the molecular weight of the unknown gas. (Use 3 sig figs and amu for the units of the answer.)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 03:00
Which step in naming unsaturated hydrocarbons is used for alkenes but not alkynes
Answers: 2

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3
You know the right answer?
If 11.50 mL of an unknown gas effuses through a small hole in a heated vessel in the same time it ta...
Questions


Social Studies, 23.10.2021 22:20

SAT, 23.10.2021 22:20



Biology, 23.10.2021 22:20


Mathematics, 23.10.2021 22:20

Health, 23.10.2021 22:20




Mathematics, 23.10.2021 22:20



Social Studies, 23.10.2021 22:20




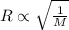
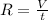
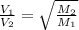
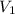 = rate of effusion of unknown gas = 11.50 mL
= rate of effusion of unknown gas = 11.50 mL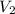 = rate of effusion of argon gas = 9.63 mL
= rate of effusion of argon gas = 9.63 mL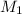 = molar mass of unknown gas
= molar mass of unknown gas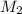 = molar mass of argon gas = 40 amu
= molar mass of argon gas = 40 amu