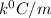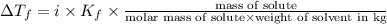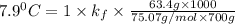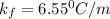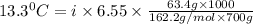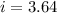Chemistry, 07.04.2020 19:35 rachelkim999
When 63.4 g of glycine (C2HNO2 are dissolved in 700. g of a certain mystery liquid X, the freezing point of the solution is 7.9 °C lower than the freezing point of pure X. On the other hand, when 63.4 g of iron(III) chloride are dissolved in the same mass of X, the freezing point of the solution is 13.3 °C lower than the freezing point of pure X Calculate the van't Hoff factor for iron(III) chloride in X.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Which substance absorbs 58.16 kj of energy when 3.11 mol vaporizes? a)ch4 b)h2s c)co2 d)nacl
Answers: 2

Chemistry, 21.06.2019 20:50
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3
You know the right answer?
When 63.4 g of glycine (C2HNO2 are dissolved in 700. g of a certain mystery liquid X, the freezing p...
Questions


Mathematics, 05.07.2019 06:20

Health, 05.07.2019 06:20


Mathematics, 05.07.2019 06:20

SAT, 05.07.2019 06:20


Mathematics, 05.07.2019 06:20





History, 05.07.2019 06:20


Chemistry, 05.07.2019 06:20

Mathematics, 05.07.2019 06:20


Mathematics, 05.07.2019 06:20

History, 05.07.2019 06:20

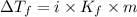
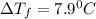 = Depression in freezing point
= Depression in freezing point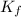 = freezing point constant =
= freezing point constant =