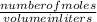Chemistry, 07.04.2020 23:03 liyahsmith9305
3. At the beginning of a titration to standardize the NaOH solution, Student A adjusted very carefully the initial burette volume to 0.00 mL, but he did not notice an important air bubble in the tip of the burette. At the end of the titration, the air bubble is gone. Explain the effect of that mistake on the calculated molarity MB. (Will the experimental MB calculated by Student A be higher or lower than the true MB value

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Write skeleton equations for the following reactions c. aluminum(s)+copper(i) chloride(aq) > aluminum chloride(aq)+copper(s)
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

You know the right answer?
3. At the beginning of a titration to standardize the NaOH solution, Student A adjusted very careful...
Questions




Health, 24.09.2019 08:30

History, 24.09.2019 08:30

Computers and Technology, 24.09.2019 08:30

Mathematics, 24.09.2019 08:30


Mathematics, 24.09.2019 08:30


Mathematics, 24.09.2019 08:30




Chemistry, 24.09.2019 08:30


Chemistry, 24.09.2019 08:30


Social Studies, 24.09.2019 08:30

Chemistry, 24.09.2019 08:30