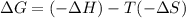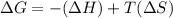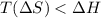Chemistry, 08.04.2020 02:29 omojay1257
For the reaction CH4(g) + 2O2(g)CO2(g) + 2H2O(g) H° = -802 kJ and S° = -5.20 J/K At standard conditions, this reaction would be product favored at all temperatures. at relatively high temperatures. at relatively low temperatures. at no temperature.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
For the reaction CH4(g) + 2O2(g)CO2(g) + 2H2O(g) H° = -802 kJ and S° = -5.20 J/K At standard conditi...
Questions

History, 01.08.2019 06:50


Biology, 01.08.2019 06:50



History, 01.08.2019 06:50

English, 01.08.2019 06:50

History, 01.08.2019 06:50

Mathematics, 01.08.2019 06:50

Mathematics, 01.08.2019 06:50

History, 01.08.2019 06:50

History, 01.08.2019 06:50

History, 01.08.2019 06:50

Mathematics, 01.08.2019 06:50



Mathematics, 01.08.2019 06:50


History, 01.08.2019 06:50

Mathematics, 01.08.2019 06:50

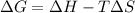
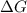 = Gibb's free energy change
= Gibb's free energy change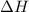 = enthalpy change
= enthalpy change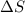 = entropy change
= entropy change