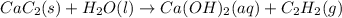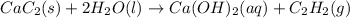Chemistry, 08.04.2020 03:13 natishtaylor1p8dirz
Solid calcium carbide, CaC2, reacts with water to form an aqueous solution of calcium hydroxide and acetylene gas, C2H2. Express your answer as a balanced chemical equation. Identify all of the phases in your answer. nothing

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1
You know the right answer?
Solid calcium carbide, CaC2, reacts with water to form an aqueous solution of calcium hydroxide and...
Questions




Social Studies, 20.09.2019 16:30


Mathematics, 20.09.2019 16:30

Chemistry, 20.09.2019 16:30

Biology, 20.09.2019 16:30





Mathematics, 20.09.2019 16:30


Social Studies, 20.09.2019 16:30



Computers and Technology, 20.09.2019 16:30

English, 20.09.2019 16:30