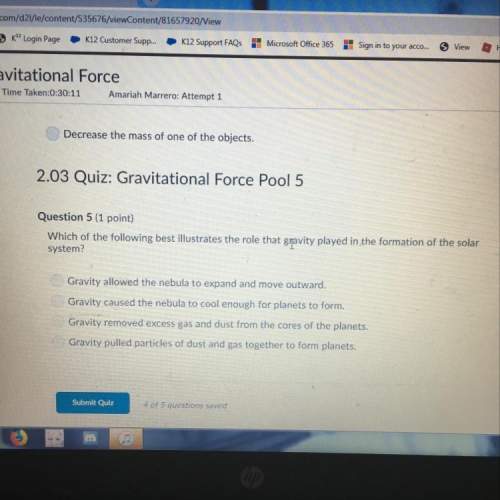
Based on Avogadro's principle, the coefficients in chemical equations involving only gases
rep...

Chemistry, 08.04.2020 04:39 dnprops1544
Based on Avogadro's principle, the coefficients in chemical equations involving only gases
represent two types of quantities. Name the two quantities.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
Questions


Mathematics, 23.04.2020 23:35

Law, 23.04.2020 23:35



Mathematics, 23.04.2020 23:35

Computers and Technology, 23.04.2020 23:35

Law, 23.04.2020 23:35

History, 23.04.2020 23:35



Social Studies, 23.04.2020 23:35



Mathematics, 23.04.2020 23:35



Mathematics, 23.04.2020 23:36

Physics, 23.04.2020 23:36

Mathematics, 23.04.2020 23:36