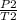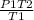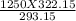Chemistry, 09.04.2020 17:48 adanaguirre17
A steel cylinder of oxygen is being store in a room at 25.0 °C under a pressure of 1250 atm. What pressure would be exerted by the gas if the container were transported across the desert at 40.0°C? SHOW WORK

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 06:30
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1
You know the right answer?
A steel cylinder of oxygen is being store in a room at 25.0 °C under a pressure of 1250 atm. What pr...
Questions


Biology, 09.07.2019 17:00

Biology, 09.07.2019 17:00








English, 09.07.2019 17:00


English, 09.07.2019 17:00

English, 09.07.2019 17:00

English, 09.07.2019 17:00

English, 09.07.2019 17:00


English, 09.07.2019 17:00

English, 09.07.2019 17:00

English, 09.07.2019 17:00

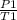 =
=