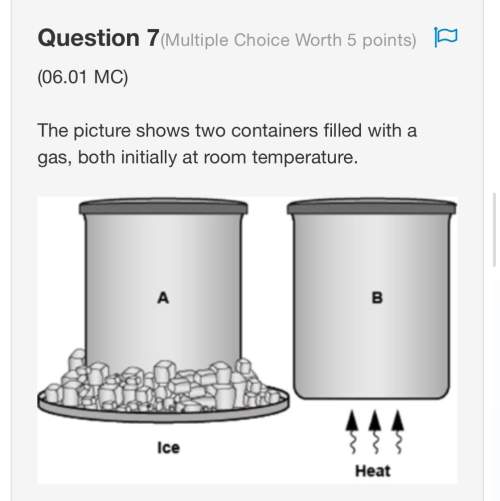
How does increasing the temperature affect a reaction?
A. An increase in temperature increases...

How does increasing the temperature affect a reaction?
A. An increase in temperature increases the reaction rate.
B. An increase in temperature slows down the reaction.
O
C. An increase in temperature lowers the reaction's activation energy.
O
D. An increase in temperature changes equilibrjum concentrations.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1

Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3
You know the right answer?
Questions



Mathematics, 11.11.2019 04:31

Mathematics, 11.11.2019 04:31




Mathematics, 11.11.2019 04:31


Mathematics, 11.11.2019 04:31

Mathematics, 11.11.2019 04:31



Chemistry, 11.11.2019 04:31

Mathematics, 11.11.2019 04:31