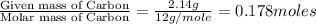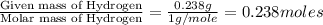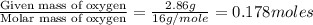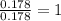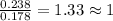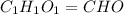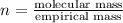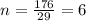Chemistry, 14.04.2020 20:14 kalebbenton15
Ascorbic acid (vitamin C) contains C, H, and O. In one combustion analysis, 5.24 g of ascorbic acid yields 7.86 g CO2 and 2.14 g H2O. Calculate the empirical formula and molecular formula of ascorbic acid given that its molar mass is about 176 g.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1


Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

You know the right answer?
Ascorbic acid (vitamin C) contains C, H, and O. In one combustion analysis, 5.24 g of ascorbic acid...
Questions


Mathematics, 24.07.2020 17:01


Computers and Technology, 24.07.2020 17:01


Mathematics, 24.07.2020 17:01



Chemistry, 24.07.2020 17:01

Mathematics, 24.07.2020 17:01

Mathematics, 24.07.2020 17:01









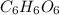
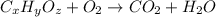
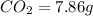
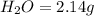
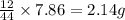 of carbon will be contained.
of carbon will be contained.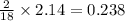 of hydrogen will be contained.
of hydrogen will be contained.