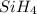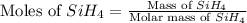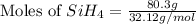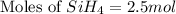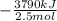Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1
You know the right answer?
The burning of 80.3 g of SiH4 at constant pressure gives off 3790 kJ of heat. Calculate △H for this...
Questions


Mathematics, 19.03.2020 00:41

History, 19.03.2020 00:41


Chemistry, 19.03.2020 00:41



English, 19.03.2020 00:41


Social Studies, 19.03.2020 00:41


Mathematics, 19.03.2020 00:41


Mathematics, 19.03.2020 00:41


Mathematics, 19.03.2020 00:41


Geography, 19.03.2020 00:41

Health, 19.03.2020 00:41

Mathematics, 19.03.2020 00:41