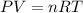Chemistry, 29.09.2019 08:30 RickandMorty420710
If the pressure, volume, and the number of moles of a gas are known, which is needed to calculate the universal gas constant from the ideal gas law?
the temperature of the gas
the molar volume of the gas
the molar mass of the gas
the partial pressure of the gas

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3


Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
If the pressure, volume, and the number of moles of a gas are known, which is needed to calculate th...
Questions

Geography, 24.04.2020 00:30

Mathematics, 24.04.2020 00:30


Mathematics, 24.04.2020 00:30


Mathematics, 24.04.2020 00:30









French, 24.04.2020 00:31




History, 24.04.2020 00:31

Health, 24.04.2020 00:31

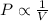 (At constant temperature and number of moles)
(At constant temperature and number of moles)
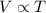 (At constant pressure and number of moles)
(At constant pressure and number of moles)
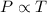 (At constant volume and number of moles)
(At constant volume and number of moles)
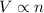 (At constant temperature and pressure)
(At constant temperature and pressure)