Relative to protons, electrons have:
A. significantly greater mass.
B. significantly le...

Chemistry, 15.04.2020 00:09 tayleeanntabeln2226
Relative to protons, electrons have:
A. significantly greater mass.
B. significantly less mass. approximately the same mass.
C. a significantly stronger positive charge.
D. None of the above is correct.
E. Electrons have no mass.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
In the analysis of hair and fiber samples, which does a compound comparison microscope allow for that a conventional compound microscope does not? a. simultaneous observation b. polarization c. fluorescence d. higher magnification
Answers: 2

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

You know the right answer?
Questions






Arts, 07.04.2020 20:32

Mathematics, 07.04.2020 20:32

Mathematics, 07.04.2020 20:32





Mathematics, 07.04.2020 20:32



Mathematics, 07.04.2020 20:32

History, 07.04.2020 20:32



History, 07.04.2020 20:32

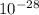 g, which is significantly lower than the mass of protons and neutrons which are approximately the same. They are extremely small (equal to 0 amu), contributing nothing to the overall mass of the atom.
g, which is significantly lower than the mass of protons and neutrons which are approximately the same. They are extremely small (equal to 0 amu), contributing nothing to the overall mass of the atom.

