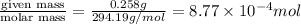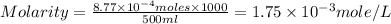Chemistry, 15.04.2020 00:18 sarinaneedshelp01
A chemist prepares a solution by adding 258 mg of K2Cr2O7 (MW = 294.19 g/mol ) to a volumetric flask, and then adding water until the total volume of the contents of the flask reaches the calibration line that indicates 500 mLmL. Determine the molarity of the prepared solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 23.06.2019 05:30
Suppose you discovered a new element with 120 protons and 2 electrons in its outer level . i'm what group does this new element belong? what properties would you expect it to have
Answers: 1

Chemistry, 23.06.2019 12:30
Choose one literary selection from this semester in which you think the setting has a great impact on the work. in a full paragraph name the work, describe the setting, and explain why it is so important to the overall story or poem.
Answers: 1

Chemistry, 23.06.2019 14:30
Which statement best identifies the process shown? the process must be fusion because energy is released. a.the process must be fusion because a heavier nucleus forms from smaller nuclei. b.the process must be fission because a large nucleus breaks into smaller nuclei. c.the process must be fission because neutrons are formed.
Answers: 1
You know the right answer?
A chemist prepares a solution by adding 258 mg of K2Cr2O7 (MW = 294.19 g/mol ) to a volumetric flask...
Questions

Mathematics, 23.02.2021 22:10

Advanced Placement (AP), 23.02.2021 22:10

Biology, 23.02.2021 22:10


Geography, 23.02.2021 22:10


Mathematics, 23.02.2021 22:10

Mathematics, 23.02.2021 22:10

Chemistry, 23.02.2021 22:10


Mathematics, 23.02.2021 22:10




Arts, 23.02.2021 22:10

History, 23.02.2021 22:10




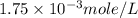
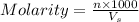
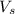 = volume of solution in ml = 500 ml
= volume of solution in ml = 500 ml