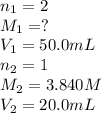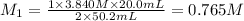Chemistry, 15.04.2020 01:00 dewillis9380
Carbonic acid, H2CO3, has two acidic hydrogens. A solution containing an unknown concentration of carbonic acid is titrated with potassium hydroxide. It requires 20.0 mL of 3.840 M KOH solution to titrate both acidic protons in 50.2 mL of the carbonic acid solution.
1. Write a balanced net ionic equation for the neutralization reaction. Include physical states.
2. Calculate the molarity of the carbonic acid solution.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 19:30
Which one of the following substances would be the most soluble in ccl4? na2so4 h2o ch3ch2ch2ch2oh c4h10 hi
Answers: 1


Chemistry, 23.06.2019 01:30
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1
You know the right answer?
Carbonic acid, H2CO3, has two acidic hydrogens. A solution containing an unknown concentration of ca...
Questions

Mathematics, 05.11.2020 18:50

Mathematics, 05.11.2020 18:50




Physics, 05.11.2020 18:50


Mathematics, 05.11.2020 18:50

Arts, 05.11.2020 18:50


History, 05.11.2020 18:50




Computers and Technology, 05.11.2020 18:50





Chemistry, 05.11.2020 18:50

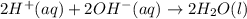
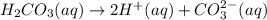 ..[1]
..[1] ..[2]
..[2]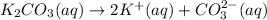 ..[3]
..[3]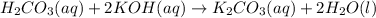
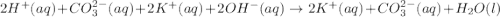
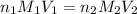
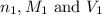 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 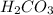
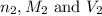 are the n-factor, molarity and volume of base which is KOH.
are the n-factor, molarity and volume of base which is KOH.