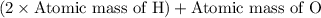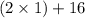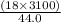Chemistry, 15.04.2020 03:07 nforrestall
A man ate 0.489 pound of cheese (an energy intake of 3.10 × 103 kJ). Suppose that none of the energy was stored in his body. What mass (in grams) of water would he need to perspire in order to maintain his original temperature? (It takes 44.0 kJ to vaporize 1 mole of water.) Enter your answer in scientific notation.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1

Chemistry, 22.06.2019 13:30
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1

Chemistry, 23.06.2019 01:30
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1
You know the right answer?
A man ate 0.489 pound of cheese (an energy intake of 3.10 × 103 kJ). Suppose that none of the energy...
Questions


Mathematics, 25.11.2020 22:50

Mathematics, 25.11.2020 22:50



English, 25.11.2020 22:50

History, 25.11.2020 22:50

History, 25.11.2020 22:50



Mathematics, 25.11.2020 22:50

Biology, 25.11.2020 22:50


Mathematics, 25.11.2020 22:50

Mathematics, 25.11.2020 22:50

History, 25.11.2020 22:50


Mathematics, 25.11.2020 22:50

Mathematics, 25.11.2020 22:50

Mathematics, 25.11.2020 22:50