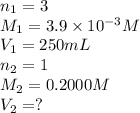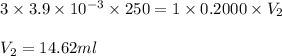Chemistry, 15.04.2020 15:52 crystalclear99
A chemistry student weighs out 0.09666 g of phosphoric acid (H3PO4), a triprotic acid, into a 250.volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.2000 M NaoH solution. Calculate the volume of NaoH solution the student will need to add to reach the final equivalence point. Round your answer to 3 significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3


Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
A chemistry student weighs out 0.09666 g of phosphoric acid (H3PO4), a triprotic acid, into a 250.vo...
Questions

Mathematics, 05.05.2020 10:44

History, 05.05.2020 10:44

Spanish, 05.05.2020 10:44

Arts, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44


English, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

History, 05.05.2020 10:44



Spanish, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

English, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

Mathematics, 05.05.2020 10:44

Biology, 05.05.2020 10:44

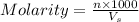
 = volume of solution in ml = 250 ml
= volume of solution in ml = 250 ml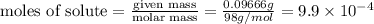
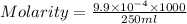
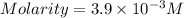
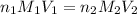
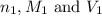 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 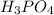
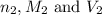 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.