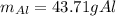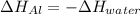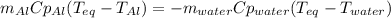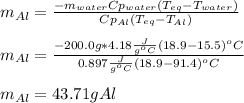Chemistry, 15.04.2020 15:49 ljcervantes4824
A chunk of aluminum at 91.4°C was added to 200.0 g of water at 15.5°C. The specific heat of aluminum is 0.897 J/g°C, and the specific heat of water is 4.18 J/g°C. When the temperature stabilized, the temperature of the mixture was 18.9°C. Assuming no heat was lost to the surroundings, what was the mass of aluminum added?
A. 243 g
B. 34.7 g
C. 41.7 g
D. 43.7 g

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1


Chemistry, 23.06.2019 06:20
What is the magnitude of the force of gravity between to 1000 kg cars which are separated by distance of 25. 0 km on an interstate highway? the force between the two cars will be what
Answers: 3
You know the right answer?
A chunk of aluminum at 91.4°C was added to 200.0 g of water at 15.5°C. The specific heat of aluminum...
Questions

Mathematics, 07.05.2020 06:59

History, 07.05.2020 06:59

Mathematics, 07.05.2020 06:59


Computers and Technology, 07.05.2020 06:59






Mathematics, 07.05.2020 06:59

Chemistry, 07.05.2020 06:59


Mathematics, 07.05.2020 06:59




Mathematics, 07.05.2020 06:59

Biology, 07.05.2020 06:59