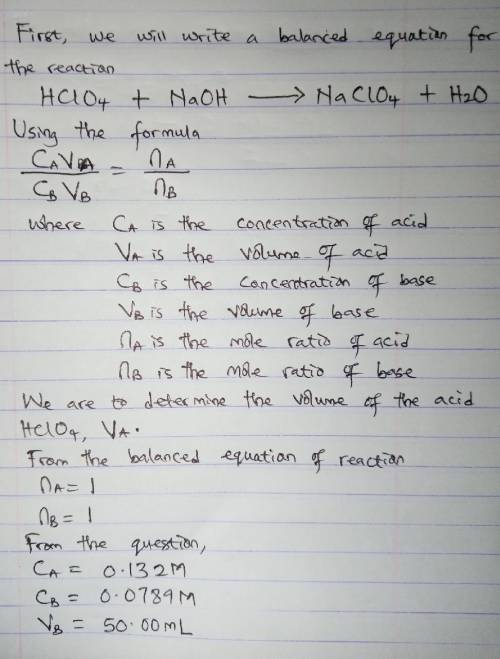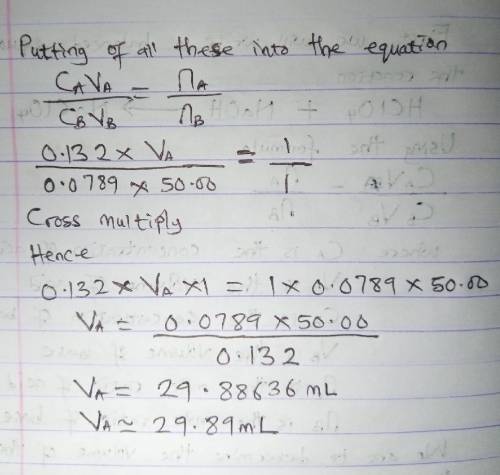Chemistry, 15.04.2020 17:30 Nathaliasmiles
How many milliliters of 0.132 m hclo4 solution are needed to neutralize 50.00 ml of 0.0789 m naoh?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
How many milliliters of 0.132 m hclo4 solution are needed to neutralize 50.00 ml of 0.0789 m naoh?...
Questions

Computers and Technology, 28.06.2019 17:00

Mathematics, 28.06.2019 17:00







Mathematics, 28.06.2019 17:00




Mathematics, 28.06.2019 17:00

Mathematics, 28.06.2019 17:00