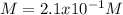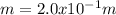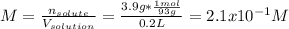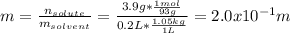Chemistry, 15.04.2020 18:54 samueldfhung
A student dissolves 3.9g of aniline (C6H5NH2) in 200.mL of a solvent with a density of 1.05 g/mL . The student notices that the volume of the solvent does not change when the aniline dissolves in it.
Calculate the molarity and molality of the student's solution. Be sure each of your answer entries has the correct number of significant digits.
How do I enter a number in scientific notation?
a. molarity =
b. molality =

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3

Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1
You know the right answer?
A student dissolves 3.9g of aniline (C6H5NH2) in 200.mL of a solvent with a density of 1.05 g/mL . T...
Questions

Mathematics, 04.01.2020 12:31

Spanish, 04.01.2020 12:31

Biology, 04.01.2020 12:31



Social Studies, 04.01.2020 12:31



Chemistry, 04.01.2020 12:31



Mathematics, 04.01.2020 12:31




Mathematics, 04.01.2020 12:31

English, 04.01.2020 12:31


History, 04.01.2020 12:31

Mathematics, 04.01.2020 12:31