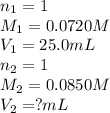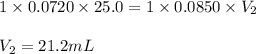Chemistry, 15.04.2020 17:46 kinglightskin2k
How many milliliters of 0.0850 m naoh are required to titrate 25.0 ml of to the equivalence point?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2

Chemistry, 22.06.2019 04:00
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
How many milliliters of 0.0850 m naoh are required to titrate 25.0 ml of to the equivalence point?...
Questions

Mathematics, 18.09.2020 22:01

Biology, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Biology, 18.09.2020 22:01

Business, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

History, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

History, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Geography, 18.09.2020 22:01

Health, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

Mathematics, 18.09.2020 22:01

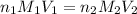
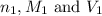 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 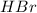
 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.