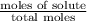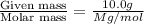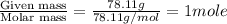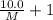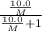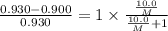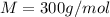Chemistry, 15.04.2020 19:55 gandalfhan
At a certain temperature, the vapor pressure of pure benzene (C6H6) is 0.930 atm. A solution was prepared by dissolving 10.0 g of a nondissociating, nonvolatile solute in 78.11g of benzene at that temperature. The vapor pressure of the solution was found to be 0.900 atm. Assuming the solution behaves ideally, determine the molar mass of the solute.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:50
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2


Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 00:30
Arrange the elements in order of increasing electronegativity. use the periodic table to you arrange the elements. p o k mg
Answers: 2
You know the right answer?
At a certain temperature, the vapor pressure of pure benzene (C6H6) is 0.930 atm. A solution was pre...
Questions

Mathematics, 24.10.2020 14:00



Geography, 24.10.2020 14:00

World Languages, 24.10.2020 14:00

Biology, 24.10.2020 14:00


Biology, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00

Mathematics, 24.10.2020 14:00


Physics, 24.10.2020 14:00

History, 24.10.2020 14:00


Mathematics, 24.10.2020 14:00

Computers and Technology, 24.10.2020 14:00


Computers and Technology, 24.10.2020 14:00

Biology, 24.10.2020 14:00

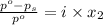
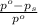 = relative lowering in vapor pressure
= relative lowering in vapor pressure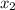 = mole fraction of solute =
= mole fraction of solute =