
Chemistry, 16.04.2020 00:50 jerenasmith77
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-8? The equation for the dissociation of hypochlorous acid is:
HOCl(aq) + H2O(l) ⇌ H3O+(aq) + OCl-(aq).

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
1) this is the structure in the cell nucleus that houses a cell's genetic information
Answers: 3


Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-...
Questions

Physics, 18.03.2021 01:00


Biology, 18.03.2021 01:00

Geography, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00

English, 18.03.2021 01:00

English, 18.03.2021 01:00



Mathematics, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00


Social Studies, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00

![[H_3O^+]](/tpl/images/0603/9545/6cfd2.png) is
is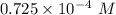 .
.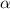 be change of concentration.
be change of concentration.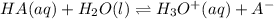
![K_a=\frac{[A^-][H_3O^+]}{[HA]}](/tpl/images/0603/9545/2421d.png)
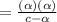
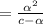
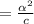 [ For weak acid , the value of
[ For weak acid , the value of 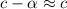 ]
]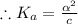
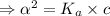
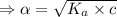
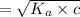
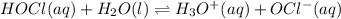
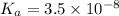
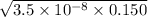 M
M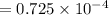 M
M

