Chemistry, 15.04.2020 21:59 chloejason8375
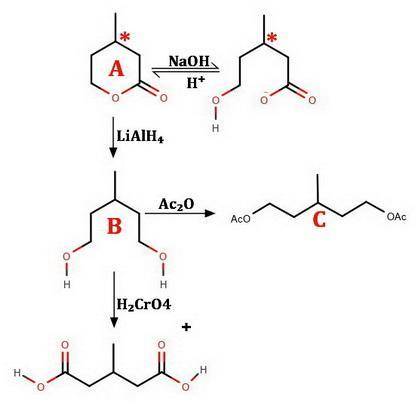
An optically active compound A, C6H10O2, when dissolved in NaOH solution, consumed one equivalent of base. On acidification, compound A was slowly regenerated. Treatment of A with LiAlH4 in ether followed by protonolysis gave an optically inactive compound B that reacted with acetic anhydride to give an acetate diester derivative C. Compound B was oxidized by aqueous chromic acid to β-methylglutaric acid (3-methylpentanedioic acid), D. Identify compounds A, B, and C; do not specify stereochemistry. (The absolute stereochemical configurations of chiral substances cannot be determined from the data.)

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
An optically active compound A, C6H10O2, when dissolved in NaOH solution, consumed one equivalent of...
Questions