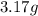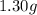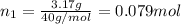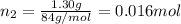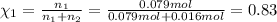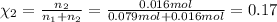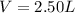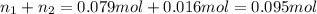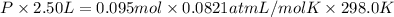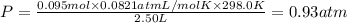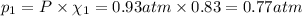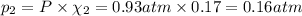Chemistry, 15.04.2020 22:05 cjjjjjjjjjjjjj
If a gaseous mixture is made by combining 3.17 g Ar 3.17 g Ar and 1.30 g Kr 1.30 g Kr in an evacuated 2.50 L container at 25.0 ∘ C, 25.0 ∘C, what are the partial pressures of each gas, P Ar PAr and P Kr , PKr, and what is the total pressure, P total , Ptotal, exerted by the gaseous mixture?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 23.06.2019 10:30
Describe the hybridization of each carbon and nitrogen atom in each of the following structures
Answers: 1
You know the right answer?
If a gaseous mixture is made by combining 3.17 g Ar 3.17 g Ar and 1.30 g Kr 1.30 g Kr in an evacuate...
Questions

Geography, 06.10.2019 01:50

Social Studies, 06.10.2019 01:50

English, 06.10.2019 01:50

French, 06.10.2019 01:50

Mathematics, 06.10.2019 01:50

Computers and Technology, 06.10.2019 01:50

English, 06.10.2019 01:50

Mathematics, 06.10.2019 01:50

Mathematics, 06.10.2019 01:50


Biology, 06.10.2019 01:50






Health, 06.10.2019 01:50