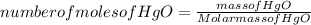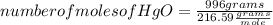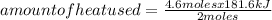Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1
You know the right answer?
A mercury mirror forms inside a test tube as a result of the thermal decomposition of mercury(ii) ox...
Questions


English, 13.02.2020 01:43

Mathematics, 13.02.2020 01:43


Mathematics, 13.02.2020 01:43


Mathematics, 13.02.2020 01:43

Mathematics, 13.02.2020 01:43

Mathematics, 13.02.2020 01:43



Computers and Technology, 13.02.2020 01:43



English, 13.02.2020 01:44




Mathematics, 13.02.2020 01:44

Mathematics, 13.02.2020 01:45