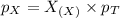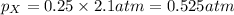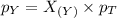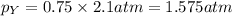A gas cylinder contains 2.0 mol of gas X and 6.0 mol of gas Y at a total pressure of 2.1 atm. What is the partial pressure of gas Y? Use StartFraction P subscript A over P subscript T EndFraction equals StartFraction n subscript a over n subscript T EndFraction..

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1
You know the right answer?
A gas cylinder contains 2.0 mol of gas X and 6.0 mol of gas Y at a total pressure of 2.1 atm. What i...
Questions





Computers and Technology, 06.08.2019 00:30

History, 06.08.2019 00:30


Mathematics, 06.08.2019 00:30








Mathematics, 06.08.2019 00:30


Engineering, 06.08.2019 00:30


Mathematics, 06.08.2019 00:30

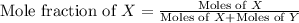
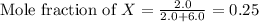
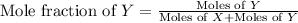
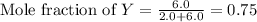
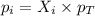
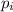 = partial pressure of gas
= partial pressure of gas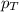 = total pressure of gas = 2.1 atm
= total pressure of gas = 2.1 atm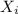 = mole fraction of gas
= mole fraction of gas