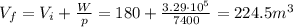Chemistry, 18.04.2020 20:09 georgettemanga2001
A cell contains 180 m^3 of gas at 7400 Pa and a machine. The machine is turned on remotely and expands the box. During this process, the machine also gives off 260 kJ of heat to the gas, and the internal energy is determined to be -69kJ. What is the final volume of the cell? Assume pressure stays constant.
Please Show All Work

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
A cell contains 180 m^3 of gas at 7400 Pa and a machine. The machine is turned on remotely and expan...
Questions


Mathematics, 10.11.2020 05:10

Mathematics, 10.11.2020 05:10


English, 10.11.2020 05:10

Mathematics, 10.11.2020 05:10

English, 10.11.2020 05:10

French, 10.11.2020 05:10

Mathematics, 10.11.2020 05:10



Mathematics, 10.11.2020 05:10

Mathematics, 10.11.2020 05:10

Chemistry, 10.11.2020 05:10


History, 10.11.2020 05:10




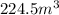

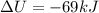 is the change in internal energy of the gas
is the change in internal energy of the gas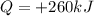 is the heat absorbed by the gas
is the heat absorbed by the gas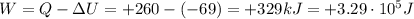
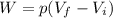
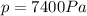 is the pressure of the gas
is the pressure of the gas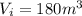 is the initial volume of the gas
is the initial volume of the gas is the final volume
is the final volume