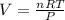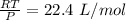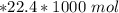The molecular weight of hemoglobin is 64,500 daltons (= g mol-1).
a. how much oxygen can...

Chemistry, 20.04.2020 20:03 finnierunners2
The molecular weight of hemoglobin is 64,500 daltons (= g mol-1).
a. how much oxygen can be bound per g of hemoglobin? express the values as ml per g of hemoglobin, where ml is the volume of o2. r =0.082 l atm mol-1 ok-1, ok =273 + oc. the molar volume of o2 is given as v = nrt/p where p = 1 atm and n= 1 mol; (hint: 1.0 mol x 0.082 l atm mol-1 ok-1 x 273.2 ok / 1 atm = 22.4 l mol-1)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1
You know the right answer?
Questions


Mathematics, 12.03.2021 20:00



Mathematics, 12.03.2021 20:00

Mathematics, 12.03.2021 20:00

English, 12.03.2021 20:00










Mathematics, 12.03.2021 20:00

English, 12.03.2021 20:00


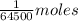
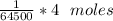
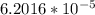 moles
moles