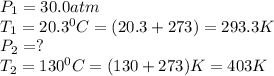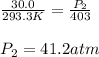A gas is placed in a storage tank at a pressure of 30.0 atm at 20.3 C. As a safety device, a small metal plug in the tank is made of a metal alloy that melts at 130 C. If the tank is heated, what is the maximum pressure (in atm) that will be attained in the tank before the plug melts and releases gas?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:10
The peak wavelength for the blackbody curve of a star is in the uv range. assuming the radiation from this star can reach earth, would you be able to see it?
Answers: 2

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3
You know the right answer?
A gas is placed in a storage tank at a pressure of 30.0 atm at 20.3 C. As a safety device, a small m...
Questions

Mathematics, 24.05.2021 20:10



Computers and Technology, 24.05.2021 20:10


Mathematics, 24.05.2021 20:10

Advanced Placement (AP), 24.05.2021 20:10

Mathematics, 24.05.2021 20:10

Computers and Technology, 24.05.2021 20:10




Mathematics, 24.05.2021 20:10

Spanish, 24.05.2021 20:10


History, 24.05.2021 20:10



Mathematics, 24.05.2021 20:10

Mathematics, 24.05.2021 20:10

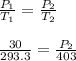
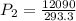
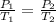
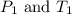 are the initial pressure and temperature of the gas.
are the initial pressure and temperature of the gas.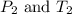 are the final pressure and temperature of the gas.
are the final pressure and temperature of the gas.