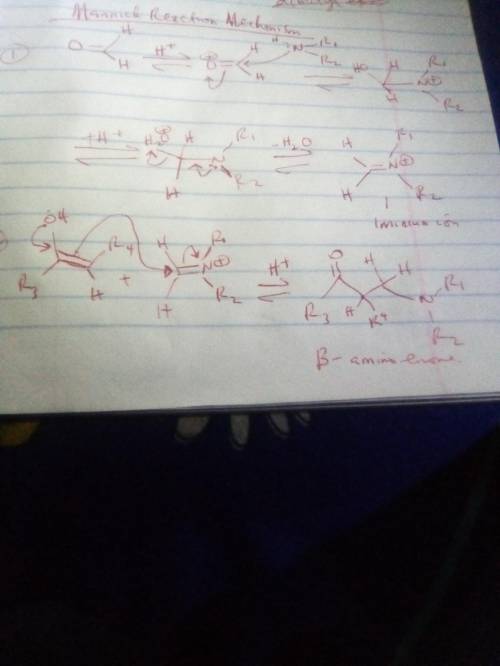
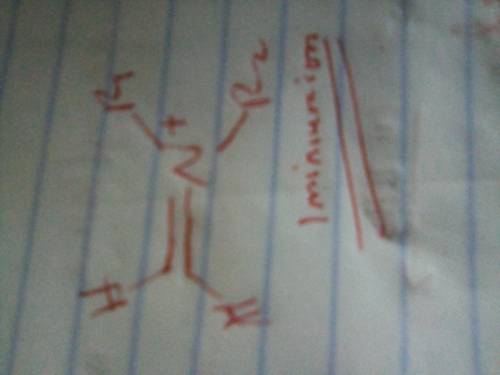
The Mannich reaction is one of the few three-component reactions in organic chemistry. In this reaction, a ketone, an aldehyde and an amine react together under acid catalyzed conditions to form the final product. The mechanism involves the following steps: 1. Following initial protonation of the carbonyl oxygen, nucleophilic attack by the amine forms a protonated carbinolamine 1; 2. Proton transfer and elimination of water forms iminium ion 2; 3. The enol form of the ketone attacks the iminium ion to form adduct 3; 4. Deprotonation of adduct 3 leads to the final product. Write out the mechanism on a separate sheet of paper and then draw the structure of iminium ion 2.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1

Chemistry, 23.06.2019 01:30
Polar bears give birth and hunt on sea ice. which of the following would polar bears survive during the melting of arctic ice? growing another layer of fur during summer migrate inland to search for different food sources staying put until the ice refreezes sticking to the usual diet of seals
Answers: 1

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2
You know the right answer?
The Mannich reaction is one of the few three-component reactions in organic chemistry. In this react...
Questions

History, 25.07.2019 00:30



History, 25.07.2019 00:30

Spanish, 25.07.2019 00:30



Health, 25.07.2019 00:30


Spanish, 25.07.2019 00:30


Mathematics, 25.07.2019 00:30


Chemistry, 25.07.2019 00:30



English, 25.07.2019 00:30


Mathematics, 25.07.2019 00:30