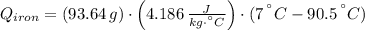Chemistry, 21.04.2020 17:37 kaylaanderson348
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 grams of water at 7.0 degrees Celsius. The final temperature of the water and the iron is 90.5 degrees Celsius. Assuming no heat is lost to the surroundings, how much heat (in Joules) does the iron release? Round your answer to the nearest 0.1 Joules.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 g...
Questions

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00


Physics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

History, 18.03.2021 01:00




Business, 18.03.2021 01:00


Mathematics, 18.03.2021 01:00


Geography, 18.03.2021 01:00

History, 18.03.2021 01:00




Mathematics, 18.03.2021 01:00